
Polar and Nonpolar Molecules
Nitrate [NO3]- is a non-polar molecular ion. It consists of a nitrogen (N) atom and three oxygen (O) atoms. The nitrogen atom is present at the center of the molecular ion, while three oxygen (O) atoms occupy terminal positions, one on each side, making a symmetrical trigonal planar shape.

Solved 2. Draw Lewis Structures For CO2, SO, And NO3 3. G...
Is NO3- Polar or Nonpolar? (Nitrate) Geometry of Molecules 2.86K subscribers Subscribe Subscribed 1 2 3 4 5 6 7 8 9 0 1 2 3 4 5 6 7 8 9 0 1 2 3 4 5 6 7 8 9 Share 664 views 1 year ago Polarity of.

Is SO3 Polar or Nonpolar? Techiescientist
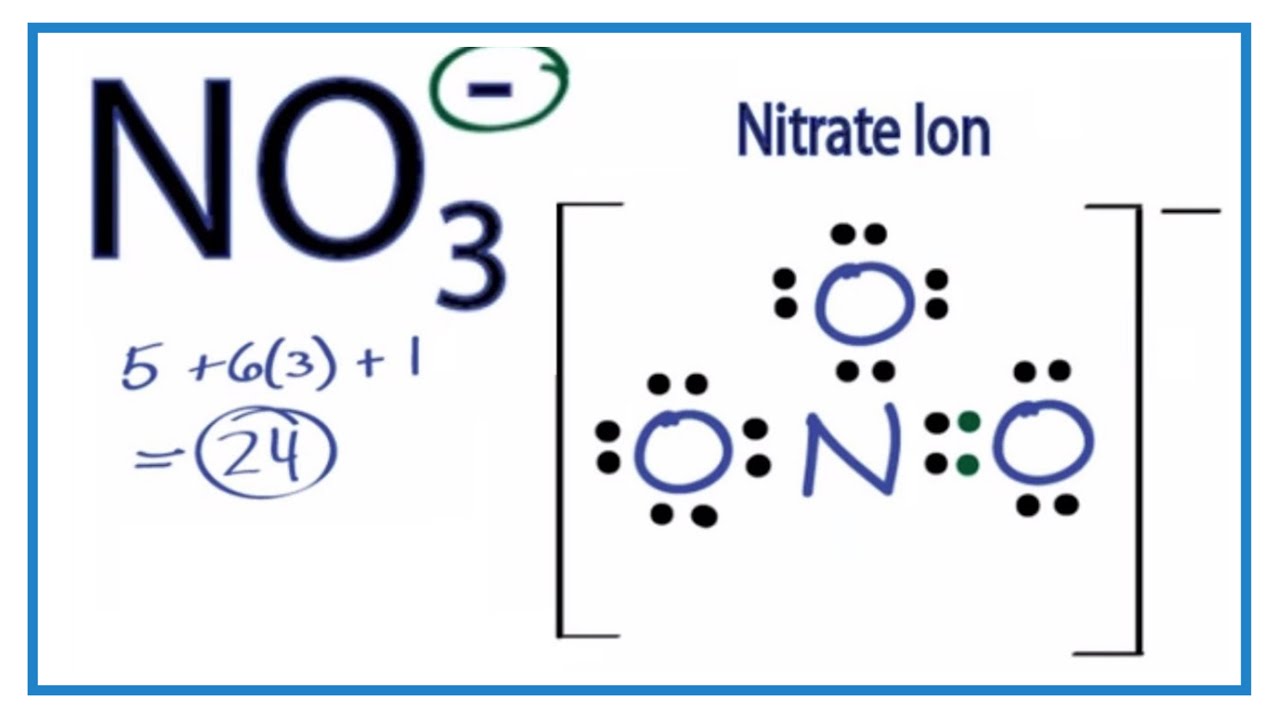
Contents show Construction of NO3 Lewis Dot Structure 1. In the ion NO3, there is 1 atom of nitrogen and 3 atoms of oxygen. It also has one negative charge. 2. Nitrogen and oxygen belong to periods 5A and 6A groups respectively in the periodic table. Hence, oxygen has 6 and nitrogen has 5 valence electrons in their outer shell.
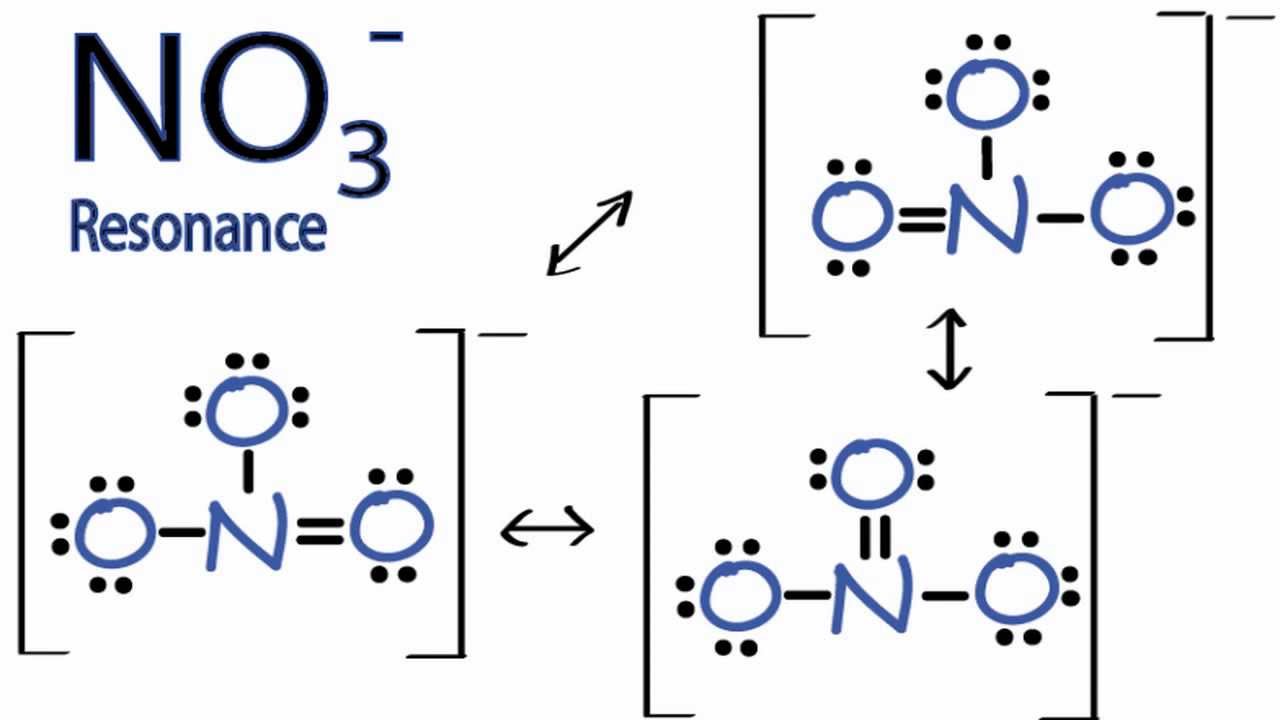
Resonance Structures for NO3 (Nitrate Ion) YouTube
Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. Electronegativity is a measure of the tendency of an atom to attract electrons (or electron density) towards itself.. {NO3-}\), and \(\ce{NH4+}\), are held together by polar covalent bonds. However, these polyatomic ions.

Is Hexane Polar or Nonpolar
It is non polar because it is a symmetrical trigonal planar structure. Explanation: I assume you mean NO3- (the nitrate anion); there is no NO3 molecule. It is non-polar because it has a trigonal planar structure and the symmetry means that there is an even distribution of electron charge density over the three N - O bonds.

Draw The Lewis Structure Of No3 Fotodtp
It is non-polar because it has a trigonal planar structure and the symmetry means that there is an even distribution of electron charge density over the three N−O bond. Whilst often draw the structure of NO3−I as a Lewis structure comparison double and single bonds, the reality is that the three N−O bonds are actually identical.

Brilliant Combustion Of Ch3oh Chemical Reaction In Fire Extinguisher
NO3 is a non-polar molecule because of the symmetrical structure irrespective of the presence of a double bond, which cancels out all the dipole moments inside the molecule. As there is no net dipole moment, there will be no separation of charges between the two ends of the molecule and hence no polarity. Contents show
is no3 polar or nonpolar
Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. Electronegativity is a measure of the tendency of an atom to attract electrons (or electron density) towards itself.. {K+}[/latex] and [latex]\ce{NO3-},[/latex] as well as covalent between the nitrogen and oxygen atoms in.

Is NO3 Polar or Nonpolar? Techiescientist
DO NOT FORGET TO SUBSCRIBE!LinkedIn: https://www.linkedin.com/in/kevan-j-e.Snapchat: https://www.snapchat.com/add/kravonoInstagram: https://www.instagram.c.

SOLVED List and know all of the names of the monatomic cations and
Unlike polar bonds, non-polar bonds share electrons equally. A bond between two atoms or more atoms is non-polar if the atoms have the same electronegativity or a difference in electronegativities that is less than 0.4. An example of a non-polar bond is the bond in chlorine. Chlorine contains two chlorine atoms.
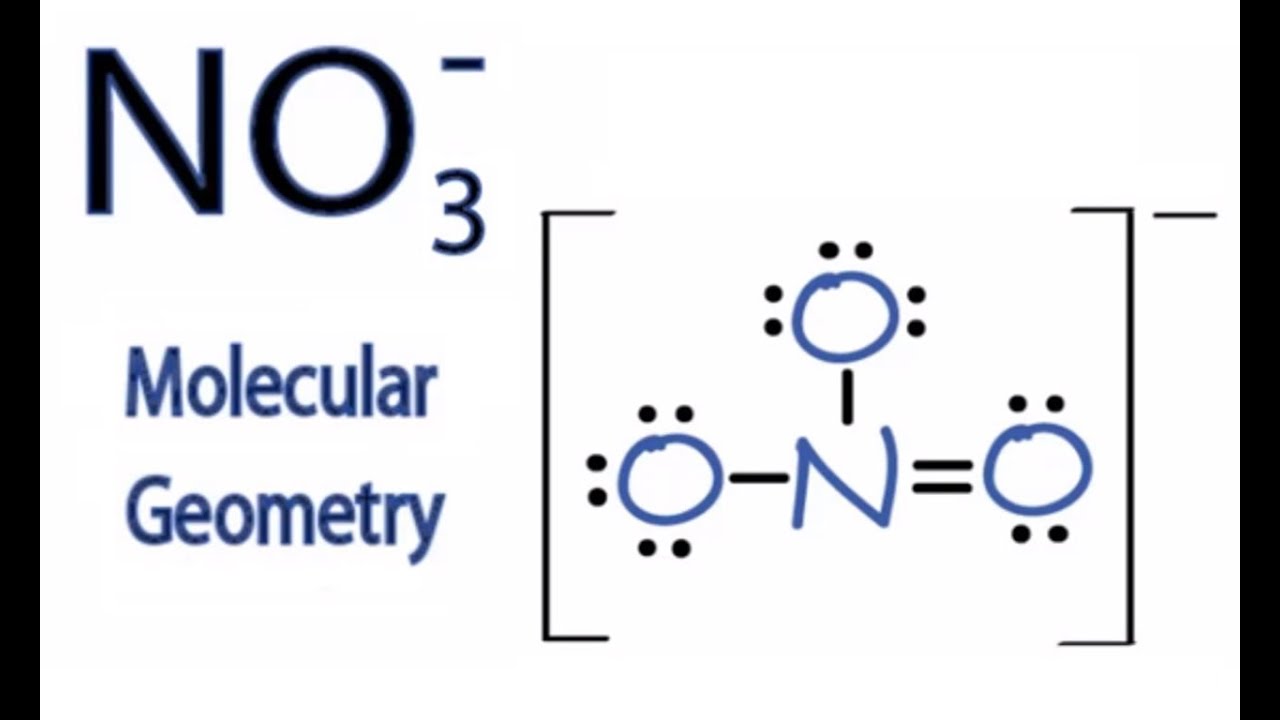
NO3 Molecular Geometry / Shape and Bond Angles YouTube
NO3- (or Nitrate ion) is a NONPOLAR ion because all the three bonds (one N=O and two N-O bonds) are equidistant and NO3- has symmetrical geometry. Let me explain this in detail with the help of NO3- lewis structure and its 3D geometry. Why is NO3- a Nonpolar ion? (Explained in 2 Steps)

is no3 polar or nonpolar
When you place a molecule with an electric dipole in an electric field, a force acts to turn the molecule so that the positive and negative ends line up with the field. The magnitude of the turning force is given by the formula. µ = q × d. where q is the amount of charge and d is the distance between the two charges. µ is the turning moment.
Workouts for weight loss for beginners, no3 shape
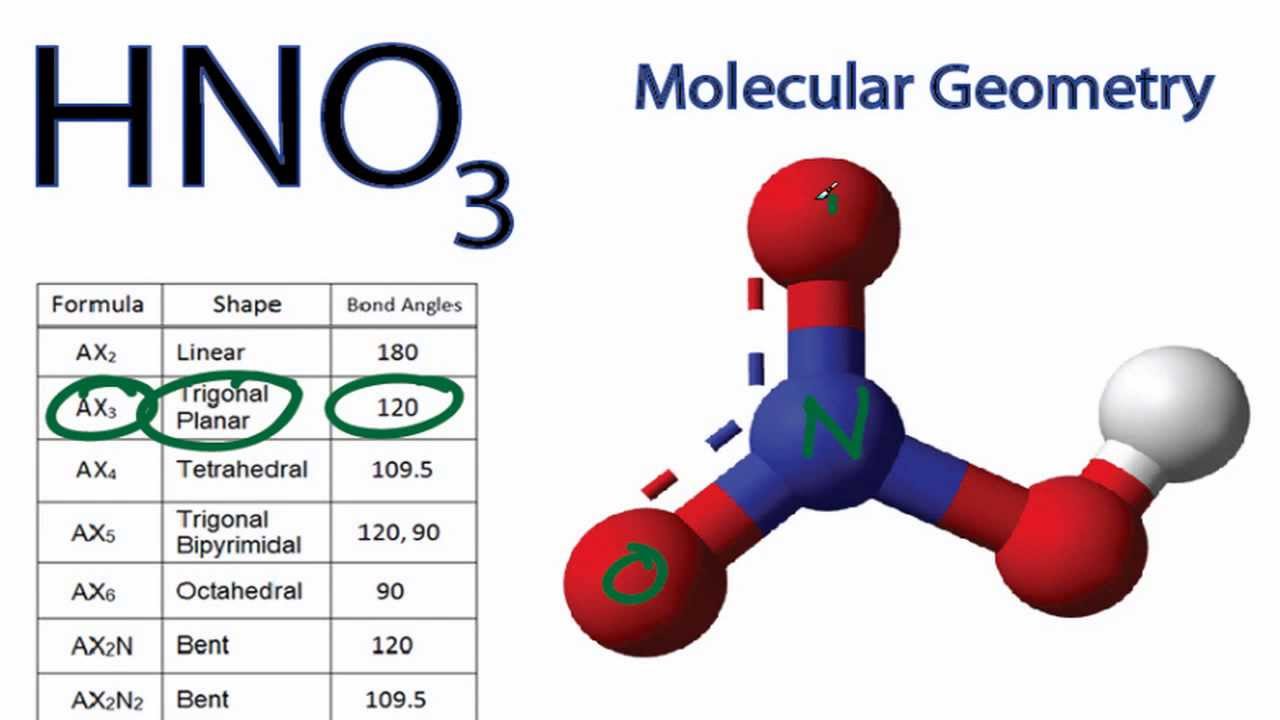
Want to know the reason? Let's dive into it! HNO3 is a POLAR molecule because the O-H bond present in the molecule is polar, which causes the partial positive (ẟ+) and partial negative (ẟ-) charge to appear on the molecule. These ẟ+ and ẟ- charges are responsible to make the entire HNO3 molecule polar.

Lewis Structure NO3 plus dipoles, shape, angles, resonance and formal
A rough approximation of the electronegativity differences associated with covalent, polar covalent, and ionic bonds is shown in Figure 3.5.0.4 3.5.0. 4. This table is just a general guide, however, with many exceptions. For example, the H and F atoms in HF have an electronegativity difference of 1.9, and the N and H atoms in NH 3 a difference.

Lectura Enlaces covalentes Biología I Association LEA
Is NO3- Polar or Nonpolar? (Nitrate ion) Wayne Breslyn 716K subscribers Subscribe Subscribed 15K views 2 years ago Learn to determine if NO3- is polar or nonpolar based on the polarity.

So2 Polar Or Nonpolar / A) Draw lewis structures for CO2, SO2, AND NO3
Molecular Polarity. To determine if a molecule is polar or nonpolar, it is frequently useful to look at Lewis structures. Nonpolar compounds will be symmetric, meaning all of the sides around the central atom are identical - bonded to the same element with no unshared pairs of electrons.Polar molecules are asymmetric, either containing lone pairs of electrons on a central atom or having atoms.