
Delta Ashlee
What is the name of this formula? Google could not provide me with an answer. ΔE = q + w Δ E = q + w physical-chemistry thermodynamics Share Cite Improve this question Follow this question to receive notifications edited Aug 11, 2014 at 5:15 Martin - マーチン ♦ 43.8k 13 13 gold badges 157 157 silver badges 314 314 bronze badges

Delta Logo White TechCrunch
The First Law of Thermodynamics states that energy can be converted from one form to another with the interaction of heat, work and internal energy, but it cannot be created nor destroyed, under any circumstances. Mathematically, this is represented as. ΔU = q + w (1) (1) Δ U = q + w. with. ΔU Δ U is the total change in internal energy of a.

Derive the relationship between Delta H and Delta U Given N(2)(g)
1 Heat is the total kinetic energy of all atoms of the system. When work is done on the system it means that a part of system kinetic energy is used to do the work, and this work makes the surrounding warmer. So " ΔU Δ U " of the system is equal to " Q Q ". And now, why we use the work of the system in: ΔU = Q + W Δ U = Q + W? physical-chemistry

FileDelta Air Lines B767332ER N394DL.jpg Wikimedia Commons
4 The difference in sign in the two versions of the first law of thermodynamics is to handle the two ways in which work can be defined. The work done (assuming only pressure-volume work) can be defined as w = PΔV w = P Δ V This is the definition often used in in scenarios when we care about the fate of the work.

(PDF) Some basic facts on the system \Delta u W_u (u) = 0
First Law of Thermodynamics The first law of thermodynamics is the application of the conservation of energy principle to heat and thermodynamic processes: . The first law makes use of the key concepts of internal energy, heat, and system work.It is used extensively in the discussion of heat engines.The standard unit for all these quantities would be the joule, although they are sometimes.

How come delta U is not equal to 0 in an isotherm expansion with a van der Waals gas but it is
The former, used primarily in physics assign a positive sign to the work done by the system while the latter assigns positive sign to the work done on the system. Hence, according to the convention youe are following, the form of the First law of thermodynamics will change:- Q = ΔU + W (Clausius convention) Q = Δ U + W (Clausius convention)

9 Example Find delta U & delta S of Gas mixture YouTube
Physics: Viewer's Request: Thermodynamics #3: Why Do We Use (delta)U=Q-W and (delta)U=Q+W ? - YouTube Visit http://ilectureonline.com for more math and science lectures!To.

Conflict Meditation Resources — Delta Kappa Delta
Heat Q Work added to the system Q>0 taken away from the system Q<0 (through conduction, convection, radiation) done by the system onto its surroundings W>0 done by the surrounding onto the system W<0 Energy change of the system is Q + (-W) or Q-W Gaining energy: +; Losing energy: - 19-2. Work Done During Volume Changes Area: A Pressure: p

एक विलगित निकाय के लिए Delta U=0 इसके लिए Delta S क्या होगा
FILE - A Delta Airlines plane flies into Portland International Airport in Portland, Ore., Monday, July 20, 2009. A former Delta Airlines pilot accused of threatening to shoot a commercial airline captain if they diverted their flight to give a passenger medical attention is will make his first court appearance in Utah, Thursday, Jan. 4, 2024.
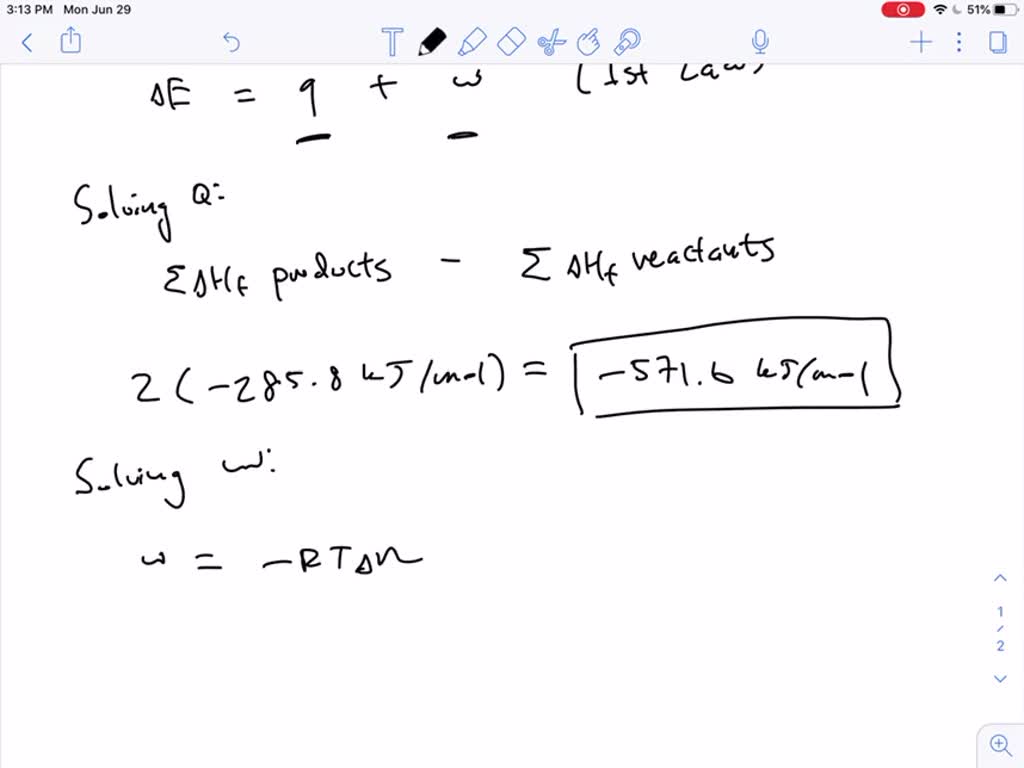
SOLVEDCalculate the ΔU for the following reaction at 298 K 2 H2(g)+O2(g) 2 H2 O(l)
The first law of thermodynamics states that the change in internal energy of a system equals the net heat transfer into the system minus the net work done by the system. In equation form, the first law of thermodynamics is. ΔU = Q − W. (15.1.1) (15.1.1) Δ U = Q − W. Here ΔU Δ U is the change in internal energy U U of the system.

FileDelta Air Lines B757351 N586NW LAX.jpg Wikipedia, the free encyclopedia
Q 1 First law of thermodynamics can be represented by ΔU = q + w. View Solution Q 2 No heat is lost or gained by the system to or from the surroundings but, work W is done on the system. Which of the following statements is correct? View Solution Q 3 A gas sample is cooled and loses 65 J of heat.

Victoria's fiveday lockdown may not quash Delta
Delta hit a low point shortly after it reported third-quarter financial results in October. The airline had a stellar summer quarter, with revenue climbing 13% to new record levels. Adjusted net.

DELTA® U494R84PK Hirsch Pipe & Supply
Δ U = Q + W [Wait, why did my book/professor use a negative sign in this equation?] Here Δ U is the change in internal energy U of the system. Q is the net heat transferred into the system—that is, Q is the sum of all heat transfer into and out of the system. W is the net work done on the system.

Relationship of delta E with q and w YouTube
Delta said it does not comment on industry speculation. While the Atlanta-based carrier placed an order for 100 Boeing (BA.N) 737 MAX 10 jets in 2022, it has primarily been an Airbus customer.

Difference Between Delta8tetrahydrocannabinol and Delta9 Tetrahydrocannabinol Delta 8 Delta 8
ΔU = Q - W Here ΔU is the change in internal energy U of the system. Q is the net heat transferred into the system —that is, Q is the sum of all heat transfer into and out of the system. W is the net work done by the system —that is, W is the sum of all work done on or by the system.

Store Delta Modifications
Delta, which has strong exposure to booming international travel, was an outperformer as the shares rose 22%—the NYSE Arca Global Airline Index rose 14% last year. Its earnings may go some way.